At medXteam, the focus is on clinical data. We collect this through literature searches or directly with the medical device as part of clinical trials. This article shows how the literature search represents an interface and how the data can be collected digitally.
Abbreviations
CEP Clinical Evaluation Plan
CER Clinical Evaluation Report
CIP Clinical investigation plan
DiGA digital health application
MDR Medical Device Regulation; EU Regulation 2017/745
PMCF Post-market clinical follow-up
SotA State of the Art
Underlying regulations
EU Regulation 2017/745 (MDR)
Medical Device Implementation Act (MPDG)
1 Introduction
Clinical data is essential for every medical device manufacturer and for every medical device:
“Clinical data” means safety or performance information obtained through the use of a product from the following sources:
- clinical trial(s) of the product in question,
- clinical trial(s) or other studies reported in the scientific literature on a product whose similarity to the product in question can be demonstrated,
- in peer-reviewed scientific literature published reports of other clinical experience either with the device in question or with a device whose similarity to the device in question can be demonstrated,
- clinically relevant information from post-marketing surveillance, in particular from post-marketing clinical follow-up.
(SOURCE: MDR, Article 2)
How the clinical data is collected should be determined at the beginning of every product development. If necessary, we collect data on behalf of the manufacturers via clinical investigations or literature searches, which in turn also serve as the basis for clinical investigation concepts, evaluation concepts for DiGA studies and descriptions of the state of the art (SotA).
The clinical data collected is summarized in the clinical evaluation or as part of its updating. The clinical evaluation is therefore at the center of every data collection or, to put it another way, every data collection flows into the clinical evaluation, which then analyzes and assesses this data.
This article shows our solution for digitally and automatically collecting clinical data for clinical evaluation or as input for a clinical trial.
2. Types of clinical trials
For medical devices, a distinction is made between four different clinical test types:
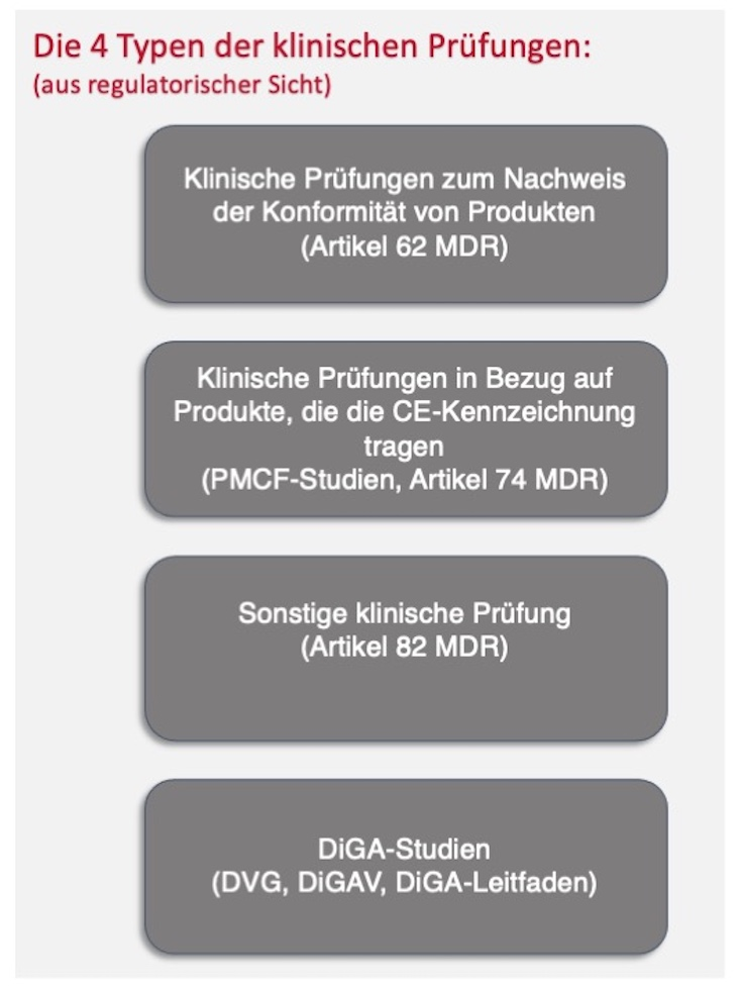
Fig. 1 Clinical examination types
The focus of this article is in particular on the clinical trial to demonstrate the conformity of products (Article 62 ff of the MDR, also called “authorization study”) and the clinical trial in relation to products that bear the CE marking (Article 74 of the MDR or exception of which, also called the “PMCF study”) as well as the DiGA studies.
Other clinical trials (Article 82 of the MDR) serve primarily to gain scientific knowledge and take place outside of the conformity assessment procedure and outside of clinical follow-up (Post Market Clinical Follow-up, PMCF). They are therefore not included in the clinical evaluation. Nevertheless, a literature search must also be carried out for test plans for these other studies.
2.1 Clinical testing to demonstrate product conformity
Such a clinical trial is also called an approval study. In this case, the clinical data is collected directly on the medical device as part of a clinical trial. In addition to state-of-the-art data, these are then included in the initial clinical evaluation of the medical device.
For such a clinical trial, a preclinical evaluation must, among other things, be submitted. This contains:
- preclinical/verification data on the product
- Data on the state of the art
In order to obtain data on the state of the art, a literature search is necessary.
This preclinical assessment also serves as the basis for the Clinical Investigation Plan (CIP), as this is a
“Description of the relevance of the clinical trial in the context of the state of the art in clinical practice”
must contain (Appendix XV, Chapter II, sentence 3.4 of the MDR).
The clinical data summarized in the clinical test report (Appendix .
The literature search focuses on clinical data on the state of the art, alternative procedures and their outcomes.
2.2 PMCF study
For clinical trials relating to products that bear the CE marking (Article 74 of the MDR) and PMCF studies excluded from it, a trial plan must also be drawn up which - unlike registration studies - is based on the most recently created clinical assessment and its possible data gaps and remaining questions.
A literature search was also carried out as part of the clinical evaluation.
2.3 DiGA studies
DiGA studies serve to demonstrate the positive healthcare effect of the digital health application (DiGA).
For this purpose, an evaluation concept drawn up in accordance with generally recognized scientific standards must be presented in advance of the fast-track procedure, which appropriately takes into account the results of the systematic data evaluation.
“In addition to a systematic literature search and evaluation, the systematic data evaluation also includes the inclusion of our own systematically evaluated data that was obtained using the DiGA.”
Therefore, in the context of a planned DiGA study, a systematic literature search must be carried out, which then also flows into the CIP of the DiGA study.
On the one hand, this literature can partly come from clinical evaluation. Since the endpoints of the DiGA study can not only cover the claims of the clinical assessment on clinical performance, safety and clinical benefit (via proof of the medical benefit of the DiGA), but can also include endpoints on patient-relevant structural and procedural improvements, recommends a separate literature search.
3. Digitized literature search
The previous section shows:
Fig. 2 Literature search in the center
The literature search can sometimes be very time-consuming and tedious. As part of the digitization of the technical documentation, the clinical assessment and therefore in particular the literature search were digitized and the process was automated.
Since medXteam focuses on clinical data, the focus is on literature searches. We implemented this process through our partner avasis as a certified Smart Expert Partner of Siemens Digital Industries Software in the areas of Teamcenter and Polarion.
3.1 Digitized literature search via Polarion
Literature search is the core process of clinical evaluation.
When searching for literature via Polarion, a direct connection is established to the database sources (e.g. directly to PubMed).
The literature search is carried out and documented in the form of the following documents:
- Literature Search and Review Plan
The literature search and review plan describes the objective search and describes the identification of publications. It includes:
- Sources of publications
- Search terms
- defined filters
- Assessment criteria and process for identified publications
- Process for analyzing the relevant publications
- Literature Search Execution Protocol
The implementation protocol provides details of the research carried out and an overview of the history of the research. It includes:
- Search queries and results used
- Deviations from the literature search and review plan
- Overview of searches carried out and search results
- Literature Review Report
The report contains a summary of the search carried out, as well as the evaluation and analysis. It includes:
- Summary of objective search execution and results
- conducted search and selection procedures for identification by other means
- Evaluation of the identified publications
- Analysis of the relevant publications (see following section)
Documentation of the analysis
The execution of objective research in Pubmed and Pubmed Central can be partially automated with digital software solutions to ensure comprehensible and reproducible research documentation and to reduce the effort required for documenting the research results. The solution used (Polarion with avaPubmed extension) offers a direct, validated interface to Pubmed and Pubmed Central.
The full text of each potentially relevant publication is read and analyzed with regard to the scope of the literature search and the relevant clinical assessment topics in the respective clinical assessment plan. The extracted statements about safety, performance, benefits, demands or state of the art are documented.
The analysis of a single "publication" is documented in the form of a single "publication evaluation" (see diagram below): The "publication" is linked to the "publication evaluation" and the evaluation is linked to the respective "clinical evaluation object" linked in the clinical evaluation plan. The following graphic explains the connection between the individual work item types:
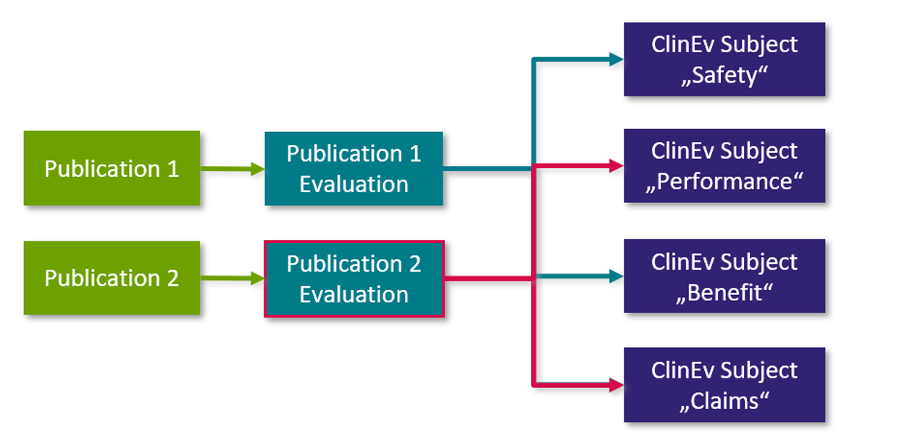
Fig. 3: Analysis
Literature search report
The literature search report provides an overview and summary of the analysis:
For each clinical assessment topic, it is listed which publication was identified as being relevant to this topic and which specific statements were extracted in the publication assessment.
Based on these results, it is analyzed whether the relevant data sets as a whole show evidence for the respective clinical evaluation subject (the respective claim, see figure above). The aim is to look for consistency of results across specific clinical assessment topics. If different results are observed across datasets, it is helpful to determine the reason for these differences.
The following graphic visualizes the connection between the documents and the digital content they contain in the form of work items:
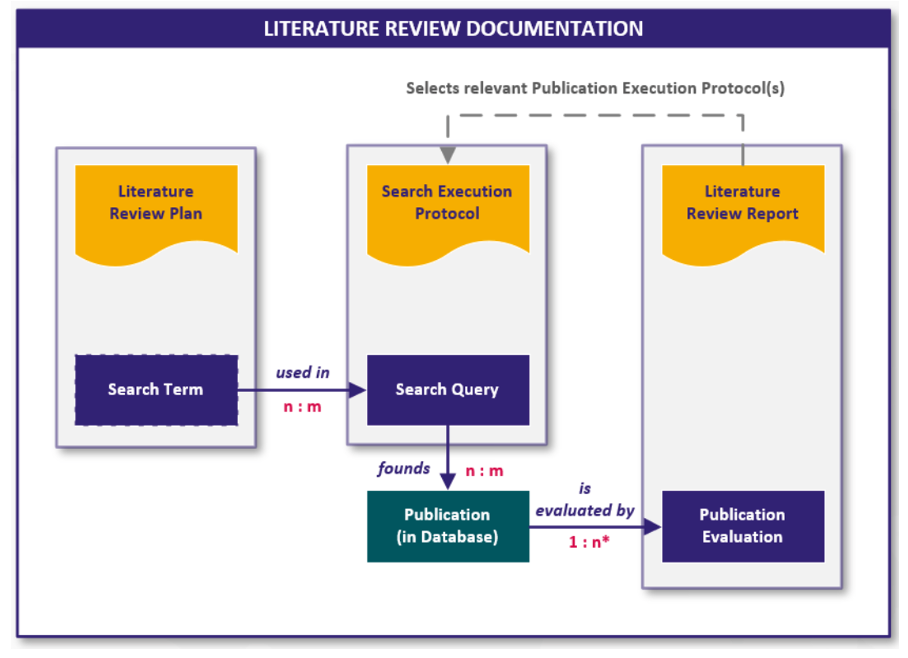
Fig. 4 Interfaces and work items
3.2 Digitalized clinical assessment
The clinical assessment is an essential part of the technical documentation (initially as part of the conformity assessment process and updates as part of the clinical follow-up).
Its core is the literature search, which can be carried out digitally (see above). Embedded in Polarion as a subsystem, it can also be digitized itself. The following figure provides an overview of the content of the clinical assessment documents
- CEP,
- CERIUM,
- Literature search documents – plan, minutes, report
embedded as a subsystem in the overall technical documentation system:
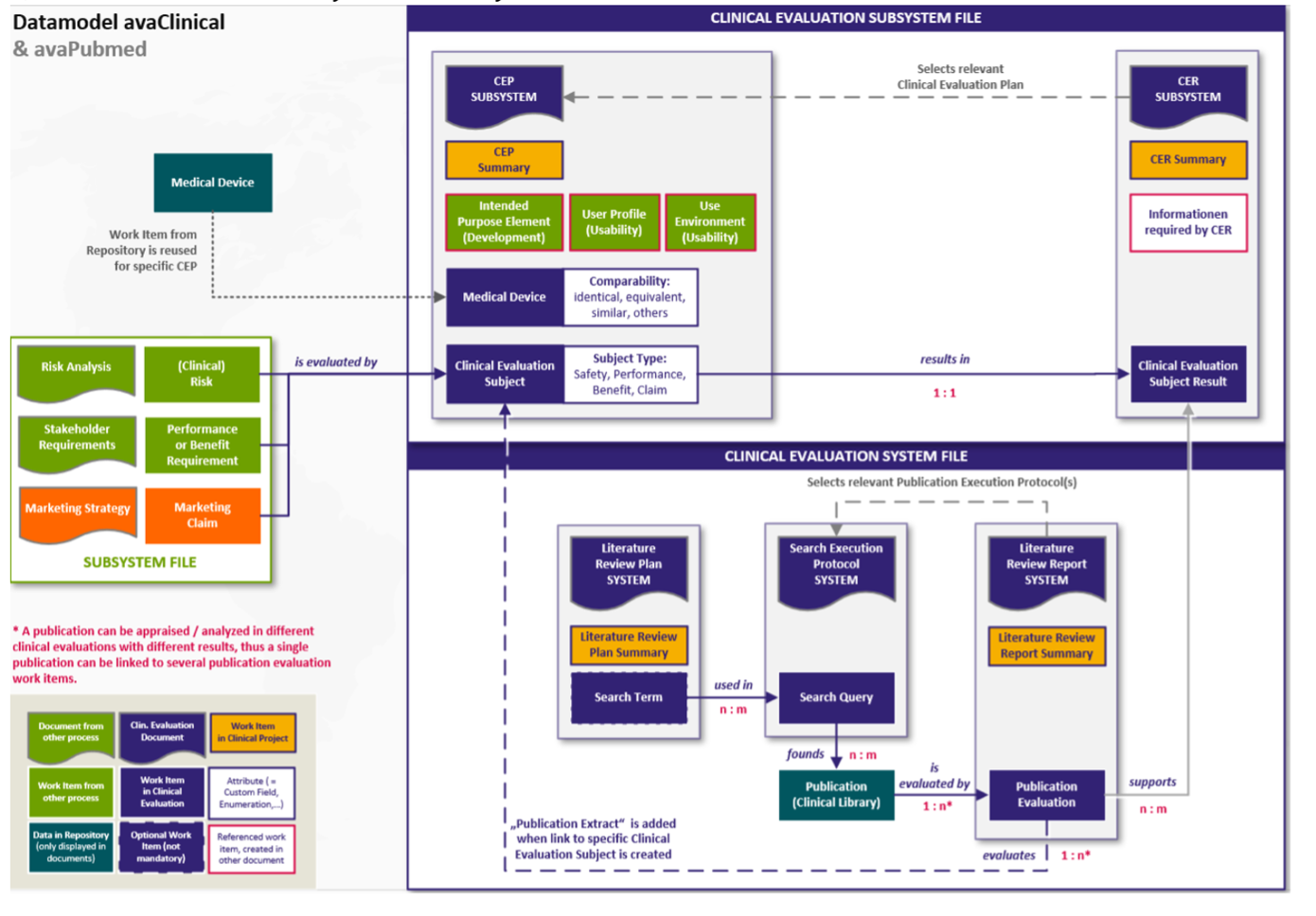
Fig. 5 Overall system technical documentation
3.3 Digitalized clinical trial
And with the close integration of the clinical trial (whether for approval, as part of the clinical evaluation or as part of a DiGA study) with the process of literature search and thus with the clinical evaluation, digitalization of the essential documents of the clinical trial, such as e.g . b.
- Clinical trial plan (Appendix XV, Chapter II, Section 3 of the MDR)
- Clinical Investigator's Manual (Appendix XV, Chapter II, Section 2 of the MDR)
- preclinical assessment
possible.
3.4 Advantages of digitalization
The digitalization of technical documentation for medical devices and thus clinical evaluation and literature search is the future!
The advantages of digitalization are obvious:
- more efficient work
- Target-oriented use of capacities
- Elimination of inefficiencies in the creation, maintenance and modification of technical documentation content, clinical evaluation and literature searches
- long-term reduction in care costs
Via Polarion, interfaces such as purpose, risk management, usability, clinical evaluation, clinical trial can be assigned to projects and reused if necessary. The creation and maintenance of documents is thus significantly simplified and accelerated. In addition, redundancies and inconsistencies are avoided.
4. What we can do for you
If a clinical trial is to be carried out, basic safety and performance requirements must first be met and essential technical documentation must therefore be created.
The clinical trial leads to the clinical evaluation, which then forms the basis for PMCF activities (including a PMCF study).
We therefore support you throughout your entire project with your medical device with primary reference to the clinical data on the product: from start to finish.
Please note two events:
October 6th, 2021, 4:00 p.m. – 8:00 p.m. Start-up Night for the healthcare industry
Question time Regulatory & Clinical Affairs – Innovation and technology meet regulation, approval and market surveillance (Hans Wenner, VDE and Daniela Penn, medXteam)
November 11th, 2021, 2:00 p.m. – 3:00 p.m.: Second free medXevent:
Digitalized clinical assessment from the perspective of clinical trials
5. How we can help you
At medXteam we clarify whether and if so which clinical trial needs to be carried out under what conditions and according to what requirements during the pre-study phase: In 3 steps we determine the correct and cost-effective strategy in relation to the clinical trial required in your case Data collection.
Do you already have some initial questions?
You can get a free initial consultation here: free initial consultation
